Hello!
A once weekly basal insulin, insulin icodec-abae, was approved by the FDA last week as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. Awiqli ("A-week-ly") is manufactured by Novo Nordisk, and availability in the U.S. is expected in coming months. Insulin icodec works by binding to serum albumin resulting in a gradual release with half-life around 1 week, thus allowing once weekly dosing. This is an important option for insulin-requiring adults with type 2 diabetes to reduce the burden of daily injections. Feel free to use the linked Awiqli Quick Tips sheet if you find it helpful.
I would love to hear your thoughts about this new FDA approval, what this means for our adult patients with T2DM and what challenges or obstacles you anticipate.
Key Information from Prescribing Information Package Insert for Awiqli:
· Awiqli is U-700, 10-unit dosing increments, max dose = 700 units
o Pen size:
§ 1050 units = 1.5 mL pen + 13 pen needles (1 pen/pack)
§ 2100 units = 3mL pen (1 pen/pack)
§ Sample Pen: 700 units = 1 mL pen + 9 pen needles
o Storage: use within 12 weeks if opened and/or stored at room temperature
o Remind patients to always use pen needle, never draw out of with syringe
· Dosing:
o New to basal insulin: recommended starting dose = 70 units once weekly
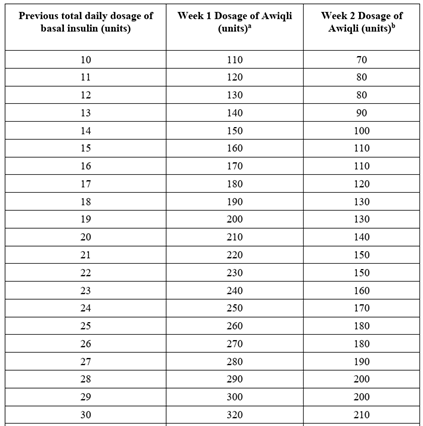
o Transition from daily basal insulin to once weekly insulin icodec-abae
-
-
- Start icodec weekly dose the day after the last daily basal dose
- See dosing table below and in package insert for prescribing information
- Week 1 dosage: 1.5 times the total daily basal dosage multiplied by 7 rounded to the nearest 10 units; Week 2 dosage = previous total daily basal insulin dose multiplied by 7 and then rounded to the nearest 10 units; continue to monitor and adjust as needed
- Steady state achieved 2-3 weeks
· Safety: Similar safety profile to other basal insulins
o Hypoglycemia, injection site reactions, allergic reactions, weight gain, hypokalemia, and HF in combination with TZDs like pioglitazone
o No apparent renal or hepatic considerations noted in clinical trials
· Efficacy: data from 4 clinical trials evaluated change in HbA1c from baseline
o ONWARDS 1 – randomized icodec once weekly vs glargine U-100 daily in adults with controlled T2DM on 1 or more oral agent (OA) or GLP1RA
§ HbA1c -1.51% icodec vs -1.33% insulin glargine (-0.18% difference, [95% CI, -0.29, -0.08]; P =.0004) = statistically significant
o ONWARDS 2 – randomized icodec once weekly vs degludec U-100 daily in adults with T2DM on once or twice daily basal insulin +/- OAs
§ HbA1c -0.90% icodec vs -0.71% degludec (-0.19% difference [95% CI, -0.32, -0.06]; P =.003) = statistically significant
o ONWARDS 3 – randomized icodec once weekly vs degludec U-100 once daily in insulin naïve adults with T2DM inadequately controlled on 1 or more OA or GLP1RA
§ HbA1c -1.56% icodec vs -1.34% degludec (-0.22% difference, [95% CI, -0.35, -0.09]; P =.0007) = statistically significant
o ONWARDS 4 – randomized icodec once weekly vs MDI (glargine + aspart)
§ HbA1c -1.16% vs 1.18% (0.02% difference) = non-inferior
See full table in Awiqli Prescribing Information Package Insert.

Reference:
Novo-Nordisk, 2026. Awiqli Prescribing Information. https://www.novo-pi.com/awiqli.pdf